BACT/ALERT® Culture Media
Provides an optimal environment for the recovery of a wide range of organisms, including bacteria, fungi and mycobacteria
- Flexible media recovers organisms from many sample types from blood to sterile body fluids
- Colorimetric technology and sophisticated algorithms minimize false negatives
- Standard and specialized Fastidious Antimicrobial Neutralization media available
- Shatter-proof plastic bottles maximize safety without compromising performance
Colorimetric detection outshines other technology
With colorimetric detection, BACT/ALERT® culture media offers you sophisticated technology that outshines the rest. Specialized Liquid Emulsion Sensors (LES) at the bottom of each culture bottle visibly change color when the pH changes due to the rise in CO2 as it is produced by microorganisms. BACT/ALERT® 3D instruments measure the color changes every ten minutes and analyze the changes. Sophisticated algorithms, including a unique “threshold” algorithm, ensure early detection of micro-organisms. Even when bottles are delayed in transit, the visual color change helps you potentially identify positive bottles prior to entry into the system. That’s how you can be assured of accurate detection and rapid recovery of microorganisms from various sample types.

- Technology and sophisticated algorithms help minimize false negatives due to delayed entry
- Low false-positive rates
- Visual, irreversible color change
Flexible media delivers reliable recovery
BACT/ALERT® culture media offers a comprehensive range of media bottles to ensure the rapid recovery of a wide variety of microorganisms including bacteria, mycobacteria, fungi, and yeasts. From various sample types including blood, sterile body fluids and sputum, BACT/ALERT® culture media can detect 98% of isolates within 72 hours*.
- FDA-cleared for blood, sterile body fluids and platelets
- Recovers a wide range of microorganisms
- Specialized bottles for mycobacterium recovery
- Fastidious Antimicrobial Neutralization media optimizes recovery for patients on antimicrobial therapy
New FAN® Plus media: optimized time to detection and recovery
Our next generation Fastidious Antimicrobial Neutralization Plus media (FAN® Plus) offers optimized time to detection and recovery. FAN® Plus bottles provide even clearer Gram stains and new Adsorbent Polymeric Beads offer antimicrobial neutralization.
- Optimized Microorganism Recovery
- Reformulated media provide a robust environment that encourages microbial growth for bacteria and yeasts
- Effective organism growth in blood and sterile body fluids
- Growth performance guidelines under delayed entry conditions
- Optimized Time To Detection
- Effective antimicrobial neutralization provides better environment for organism growth and detection
Safety & efficiency: trust the pioneer in plastic collection bottles
bioMérieux was the first to introduce revolutionary multilayer plastic bottles. Made of unique polycarbonate material, their triple-layer design ensures appropriate environment for recovery of microorganisms. These shatter-resistant bottles significantly improve safety in the laboratory by eliminating accidental spills, potential biohazard exposure and glass cuts. Because they weigh less, these bottles have the added benefit of reducing shipping and disposal costs. And – as numerous studies have shown – the bottom line is that you get all these clear advantages while still ensuring optimal performance in recovery and time to detection.
- Shatter-resistant bottles reduce potential for biohazard exposure
- Up to 2/3 lighter than glass
- Cost-effective storage, shipping and disposal
- Multi-layer, gas-impermeable design maintains aerobic or anaerobic environments
- Adapt to a variety of protocols and enable pneumatic transport without a special container

Standard Media
| Bottle Type | Media Formulation | Specimen Type | Specimen Volume | |
|---|---|---|---|---|
 |
BACT/ALERT® SA |
40 ml supplemented tryptic soy broth (TSB) |
Blood or normally sterile body fluid (SBF) |
Up to 10 ml |
 |
BACT/ALERT® SN |
40 ml TSB | Blood or SBF | Up to 10 ml |
Fastidious Antimicrobial Neutralization Plus Media (FAN® Plus)
| Bottle Type | Media Formulation | Specimen Type | Specimen Volume | |
|---|---|---|---|---|
 |
BACT/ALERT® FA Plus |
30 ml of supplemented complex media containing adsorbent polymeric beads |
Blood or normally sterile body fluid (SBF) |
Up to 10 ml |
 |
BACT/ALERT® |
40 ml of supplemented complex media containing adsorbent polymeric beads |
Blood or SBF | Up to 10 ml |
 |
BACT/ALERT® PF Plus |
30 ml of supplemented complex media containing adsorbent polymeric beads |
Blood | Up to 4 ml |
Mycobacterium Media
| Bottle Type | Media Formulation | Specimen Type | Specimen Volume | |
|---|---|---|---|---|
 |
BACT/ALERT® MP Mycobacteria Process - Ref. 259797 Use with ref. 259760 |
10 ml supplemented Middlebrook 7H9 broth. | Processed specimen or SBF other than blood. | 0.5 ml |
Platelet QC Testing
| Bottle Type | Media Formulation | Specimen Type |
Speci men Volume |
|
|---|---|---|---|---|
 |
BACT/ALERT® BPA Aerobic Platelet culture bottles – Ref. 279018 or EU Ref. 279044 |
40 ml supplemented tryptic soy broth (TSB) | Leukocyte reduced apheresis platelet (LRAP ) units, and both leukocyte reduced single and a pool of up to six (6) units of leukocyte reduced whole blood platelet concentrates (LRWBPC) | 4 – 10 ml |
 |
BACT/ALERT® BPN Anaerobic Platelet culture bottles - Ref. 279019 or EU Ref. 279045 |
40 ml supplemented tryptic soy broth (TSB) | Leukocyte reduced apheresis platelet (LRAP ) units, and both leukocyte reduced single and a pool of up to six (6) units of leukocyte reduced whole blood platelet concentrates (LRWBPC) | 4 – 10 ml |
Greiner Bio-One VACUETTE® Safety Blood Collection Sets and Bulk Adapter*
| Description | Product # | Packaging | |
|---|---|---|---|
 |
VACUETTE® Safety Blood Collection Sets Contains needle, tubing and Greiner adapter in sterilized packet. |
Ref. 413251 - Needle Size 21G, 30cm tubing. Ref. 413201 - Needle Size 23G, 30cm tubing. Ref. 415419 - Needle Size 21G, 19cm tubing. Ref. 415450 - Needle Size 23G, 19cm tubing. |
240 per case |
 |
Bulk non sterile adapter Non-sterile adapter cap with improved neck design to accommodate evacuated tubes without the need for an insert. (Female threaded Luer). |
Ref. 413200 | 320 per case |
COVIDIEN Products
| Description | Product Number | |
|---|---|---|
covidien_-_ca006.jpg Show in media library: |
MonojectTM Collection Device, Male The MonojectTM collection device is a single use device for the collection of blood specimens |
Ref. CA006 |
covidien_411350.jpg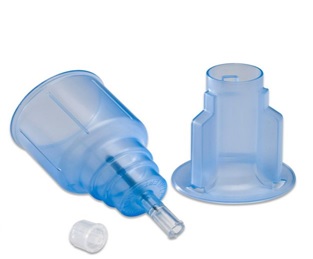 Show in media library: |
MonojectTM Transfer Device, Female The MonojectTM transfer device is single use for safe transfer of blood specimen from a syringe into a blood culture bottle |
Ref. 411350 |
Safety SubCulture Units
| Description | Product Number | |
|---|---|---|
saferty_subculture_unit.jpg Show in media library: |
Safety SubCulture Unit Safe, needleless transfer of samples from positive blood-culture bottles |
Ref. 419142 |
For product availability, please contact your local bioMérieux representative.
BACT/ALERT® VIRTUO® & BACT/ALERT® FAN® PLUS MEDIA PUBLICATIONS
BACT/ALERT® VIRTUO®
BACT/ALERT® FAN® PLUS MEDIA vs. BACTEC™
BACT/ALERT® FAN® PLUS MEDIA vs. BACT/ALERT® FAN AND STANDARD MEDIA





