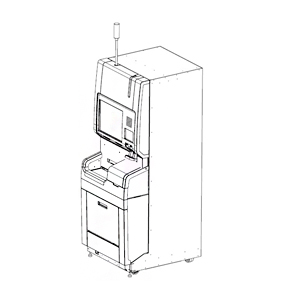
BACT/ALERT® VIRTUO®
Automated instrument for blood culture
Get ready for a new dimension in blood culture detection: the BACT/ALERT® VIRTUO® is seamlessly efficient blood culture technology in one automated system.
- Motion-activated “Set and Forget” loading
- Advanced robotics provides hands free bottle loading and unloading
- Flexible and scalable instrumentation adapts to any size laboratory
- VIRTUO® helps you redesign your workflow by reducing hands on time and increasing efficiency
BACT/ALERT® VIRTUO® ushers in a new dimension of blood culture detection. Almost endlessly versatile it fits into any size lab, even outside the routine microbiology lab. The latest example of ingenuity in automation, VIRTUO® reduces hands-on time for increased lab efficiency. These benefits can be further boosted through highly sophisticated MYLA® software that puts relevant information from multiple instruments and your LIS at your fingertips, when and where you need it. In these ways and more, BACT/ALERT® VIRTUO® seamlessly integrates your blood culture workflow to provide efficient and actionable results to help you achieve better patient outcomes.
Ultimate workflow efficiency
Your laboratory faces some critical challenges these days: higher volumes, need for fast turnaround time and the need to use staff resources effectively. Clinicians and their patients count on you meeting these challenges. To help you do that, BACT/ALERT® VIRTUO® ’s intelligent automation ensures your workflow couldn’t be easier.
- Loading area can take up to forty bottles at a time
- So straightforward that any lab staff member can load
- Automated, motion-activated “Set and Forget” loading
- Intuitive touchscreen offers enhanced ease-of-use
- Barcode issues corrected on main screen
- Advanced robotics for automated loading and unloading
- System knows when bottles are loaded
- Troubleshoots problems (e.g. a fallen bottle)
Smart scanning: rotates and photographs entire bottle.
- 360° Smart scanning technology
- Recognizes bottle and patient accession labels
- Senses and tracks blood fill level on all bottles
- Ease of tracking and reporting
- Positive bottles:
- Alerts mean you can start work without delay
- You set on-demand or automated unloading
- Negative bottles:
- Automatically unloaded into the removable waste bin
- Disposal is completely safe thanks to bioMérieux’s shatter resistant bottles
Scalability for any size laboratory
BACT/ALERT® VIRTUO® is versatile and scalable for almost any size laboratory – you can even place systems in other locations outside of the routine microbiology lab.
- Capacity from 216 to 432 cells
Improve visibility of your testing with MYLA®
The BACT/ALERT® VIRTUO® system comes with two software packages:
1 - Basic stand-alone package:
- Easy to use
- Routine reports such as Positive, Negative and Negative to date
2 - Fully integrated MYLA® middleware, so you connect to other bioMérieux instruments and your LIS to gain a “control tower” perspective to ensure information flows smoothly. This eases the administrative burden and optimizes workflow so you manage higher volumes with your existing resources and free your time for more skill-intensive work.
- Get the whole picture: information at your fingertips 24/7
- Consolidate patient data for easier and more powerful reporting
- Total traceability of blood culture workflow
- Comprehensive blood volume reporting
- Quality indicator tracking
- Improves clinical relevance with real-time statistics & quality metrics
- Contamination tracking further improved with integrated Vitek Mass Spec and Vitek 2 results
VIRTUO™ is CE marked and available in select countries globally.
Physical Specifications
- Receiving dock and the internal delivery route must be able to accommodate the size and weight of instruments being delivered.
IMPORTANT: Allow adequate space for removing the instrument from the shipping crate. The total space required to remove the instrument from the shipping crate is 200 in. (5 meters). The shipping crate ramp is 85 in. long (215.9 cm). Refer to the following Instrument and Crate dimensions:
Size:
| Height | Width | Depth | Length | |||||
|---|---|---|---|---|---|---|---|---|
| in | cm | in | cm | in | cm | in | cm | |
| Instrument (Door Closed) | 77.2* | 196.0* | 28.7 | 73.0 | 35.8 | 91.0 | ||
| Instrument (Door Open) | 77.2* | 196.0* | 37.0 | 94.0 | 65.0 | 165.0 | ||
| Shipping Crate | 89.0 | 226.1 | 36.0 | 91.4 | 48.0 | 121.9 | ||
| Shipping Crate Ramp | 85.0 | 215.9 | ||||||
*Instrument Height dimension does not include optional Warning Light Tower.
Instrument Weight:
| lb | kg | |
|---|---|---|
| Total Shipping Weight | 1084 | 491 |
| Weight of Instrument (empty) | 643 | 292 |
| Weight of Instrument (with bottles) | 707 | 320 |
| Instrument Packaging Weight | 441 | 200 |
Warning Light Tower
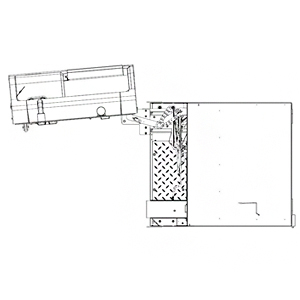
BACT/ALERT® VIRTUO® Instrument with optional Warning Light Tower
BACT/ALERT® VIRTUO® Instrument with Door Open (Top View)
2.Electrical Requirements
- Properly grounded dedicated A/C power branch circuit capable of providing:
- 20 Amps @ 100Vac, 120Vac @ 50 - 60 Hz
- 13 Amps @ 220Vac, 230Vac, 240 Vac @ 50 – 60 Hz
Note: These circuit ratings are required to accommodate the peak current draw of the instrument on a dedicated circuit
- Average Power Consumption (Watts):
- 1440 Watts (@120 VAC
- 1320 Watts (@220 VAC)
- Environmental Heat Transfer (BTU/Hr): 1287 at instrument set point of 37 °C and ambient temperature of 20°C
Notes:
- One branch circuit will provide power to the BACT/ALERT® VIRTUO® instrument.
- Each additional BACT/ALERT® VIRTUO® instrument will require a dedicated branch circuit.
- NO power strips or extension cords are to be used for power distribution to the BACT/ALERT® VIRTUO® instrument.
- AC power outlets must be within 8 ft. or 2.44 m of each BACT/ALERT® VIRTUO® instrument installed.
- Nonconformance to the above Electrical Requirements may result in voiding the equipment warranty or service contract resulting in a charge for service.
- To ensure proper operation, the supporting electrical circuit must be maintained as a dedicated power source (for example, not serving large power consuming devices including but not limited to, centrifuges, air conditioners, water baths, refrigerators, freezers, or any devices that radiate electrical noises)
- It is recommended that each BACT/ALERT® VIRTUO® instrument is connected to Emergency power branch circuits.
The BACT/ALERT® VIRTUO® instrument cannot agitate, heat, or perform bottle readings during a power outage.
3.Environmental Requirements
- Temperature Range: 59º to 82ºF (or 15º to 28ºC), with no wide temperature fluctuations
- Room Relative Humidity: 20% to 80%, non-condensing
4.Location of the BACT/ALERT® VIRTUO® Instrument
- A single Ethernet Laboratory Information System (LIS) connection must be within 20 m of the BACT/ALERT® VIRTUO® instrument if connected to an LIS.
- The BACT/ALERT® VIRTUO® instrument should not be installed under or near a HVAC register or in direct sunlight or bright overhead lights.
- The Customer must certify that the floor structure where the BACT/ALERT® VIRTUO® instrument is to be installed is capable of supporting the weight of all BACT/ALERT® VIRTUO® instruments installed, while fully loaded with culture bottles.
- To provide adequate service accessibility, a minimum unobstructed clearance of 12 in. or 30.5 cm must be provided on the left side of the BACT/ALERT® VIRTUO® instrument.
5.ViLINK Configuration
Refer to the Latest ViLINK documentation for connectivity instructions. The BACT/ALERT® VIRTUO® instrument must be connected to ViLINK in order for the instrument to be installed.
VIRTUO™ is CE marked and available in select countries globally.
For product availability, please contact your local bioMérieux representative.
BACT/ALERT® VIRTUO™ PUBLICATIONS
e-POSTER ECCMID
Cheong Y.S., Chew K.L., Jureen R. Evaluation of the BACT/ALERT VIRTUO and the BACT/ALERT 3D
Publication